EmpNia Earns FDA Clearance for Respiratory Device
Minneapolis-based medical device innovator EmpNia has been granted clearance by the Food and Drug Administration (FDA) to market its wearable sensor, eMotus, to hospitals and cancer centers across the U.S.
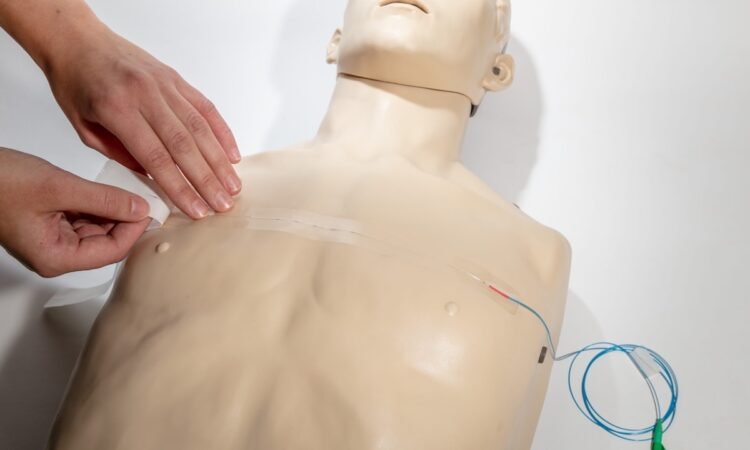
The company’s eMotus system is designed to help manage image-guided radiation therapy—specifically, for cancer patients. EmpNia’s team of medical professionals saw an opportunity to improve upon existing solutions, which they said can be hard to use, disruptive to workflow, and incompatible across various imaging and therapy modalities.
EmpNia’s system is a single-use disposable sensor pad and software that provides real-time respiratory motion tracking. Universally compatible with existing imaging and therapy equipment, eMotus enables clinics to adopt motion management without costly infrastructure investments.
“Effectively managing respiratory motion across all patient types and delivery systems, whether during imaging or radiation therapy, has long challenged clinicians and added unnecessary time and complexity. eMotus overcomes these challenges with a simple, reliable, and universally compatible solution that lets care teams stay focused on treating patients, not managing equipment,” said Dr. Cliff Robinson, EmpNia’s chief medical officer and a professor of radiation oncology and cardiology at Washington University in St. Louis. EmpNia’s founder and CEO is Manojeet Bhattacharya, who has been developing and commercializing medical devices since 2009.
In pre-market evaluations, eMotus demonstrated improvements to workflow, setup and motion tracking accuracy. Its straightforward design reduces the burden on staff and eliminates the need for maintenance contracts, Robinson said.
The system is initially intended for use by radiation oncology professionals. managing patients undergoing image-guided radiation therapy who require monitoring and management of respiratory motion during treatment. A spokesperson for EmpNia said the team sees “broad application” for eMotus down the road.