Medtronic’s Pill-Sized Pacemaker Wins FDA Approval
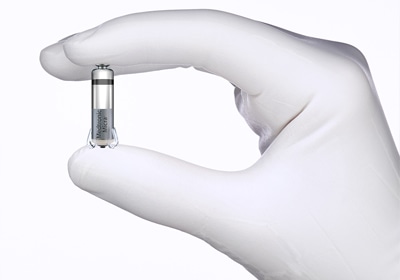
At less than one-tenth the size of traditional pacemakers, Medtronic’s Micra Transcatheter Pacing System (TPS) is the world’s smallest pacemaker and, as of Wednesday, the heart-monitoring device can now be sold in the United States.
The U.S. Food and Drug Administration (FDA) granted Medtronic regulatory approval to sell its Micra TPS pacemaker after months of trials on patients.
“For many years we’ve been hopeful that a transcatheter pacing solution—with a safety and effectiveness profile on par with conventional devices—would become available, and today Micra has achieves this milestone,” Dwight Reynolds, principal investigator of the Micra TPS global clinical trial, said in a statement. “In the clinical trial, the Micra was successfully implanted in nearly all patients, and met its safety and effectiveness endpoints by wide margins. This gives us great confidence that this miniaturized device will bring patients the most advanced pacing technology, combined with the less-invasive nature of the new technology.”
During the clinical testing period, Reynolds previously noted the Micra TPS had 51 percent fewer major complications compared to conventional pacing systems. Altogether, the device was successfully implanted in 99.2 percent of 744 patients with zero dislodgements reported.
The Micra TPS is the first-ever leadless pacemaker to enter the U.S. market. Traditional pacemakers attach to the heart with cardiac wires called leads, whereas the Micra TPS is delivered through a catheter and implanted directly into the heart.
“As the first leadless pacemaker, Micra offers a new option for patients considering a single chamber pacemaker device, which may help prevent problems associated with the wired leads,” said William Maisel, director of the office of device evaluation for the FDA’s center for devices and radiological health.
Dublin-based Medtronic, which maintains its U.S. corporate headquarters in Fridley, received approval for sales in the European Union exactly one year ago.
Not far behind Medtronic is St. Jude Medical with its own tiny leadless pacemaker, Nanostim. The Little Canada-based company received European approval for Nanostim in October 2013 and is currently in pursuit of FDA approval.
The most recent study from the American Heart Association found that roughly 3 million people worldwide use pacemakers, and each year 600,000 more pacemakers are implanted.