Eden Prairie Company Expands Med Tech Use to Chronic Pain Disorder
In March, NeuroOne Medical Technologies Corporation announced the completion of its limited market release of a medical device treating a rare pain disorder.
Trigeminal neuralgia is an often debilitating chronic pain condition affecting approximately 150,000 patients in the United States, according to NeuroOne. Harvard Health Publishing reports the disorder newly affects four to five out of 100,000 people in the country each year. It involves “sudden, intense facial pain that disrupts even the most basic daily functions,” per NeuroOne.
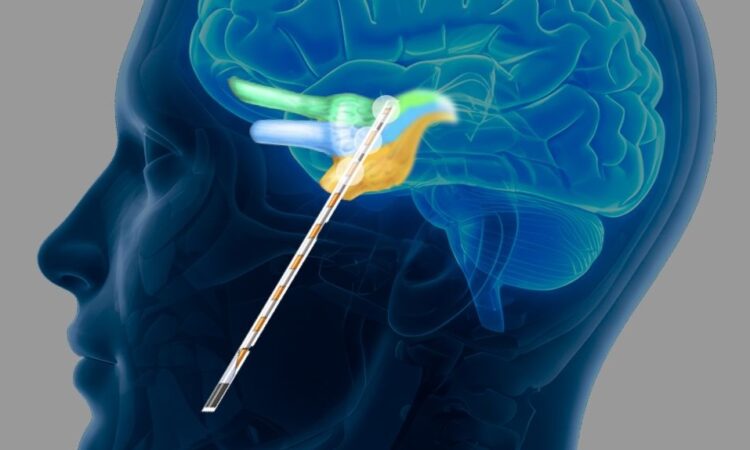
NeuroOne’s device is called the OneRF trigeminal nerve ablation system. The FDA cleared the device in August.
Twelve patients from three medical centers were treated as a part of the limited market release, and the company reports a 100% success rate. The fastest procedure was completed in 16 minutes, the company says.
“I want to get 10 cases done before I ever do a full launch of any device,” says Dave Rosa, president and CEO—to guarantee that it works and avoid a potential recall. “Once you recall a device, you know you’re in trouble.”
The trigeminal system is an expansion of the company’s OneRF product, launched in 2023 and aimed at diagnosing and treating neurological disorders. The core of NeuroOne’s technology is paper-thin electrodes, which had first been developed to treat epilepsy, Rosa says.

Origin of NeuroOne
The standard treatment for epilepsy has been craniotomy. This surgical procedure entails the removal of the top part of the patient’s skull to access the brain and find where the seizures are originating. That brain tissue is removed.
“And the end result might be, ‘We can’t treat you,’” says Rosa, noting the high risk.
Mayo Clinic, in Rochester, was looking for a less invasive procedure for epilepsy. Research at the University of Wisconsin led to the 2017 launch of NeuroOne in collaboration with Mayo, which purchased an 11% ownership stake, making the clinic the company’s first investor.
The concept was something very thin and flexible that could be inserted without having to remove the top part of the skull. The OneRF brain ablation system was cleared in December 2023 and commercially launched in March 2024.
“It’s the only system that, in essence, finds the area in the brain that’s triggering the seizures,” explains Rosa. “With the same device, when you connect it to a different piece of equipment, you can then destroy that tissue, with the expectation that you’ll be able to stop the patient’s seizures.”
What the company leaders did not know at the time was that the concept could be applied to other parts of the body.
“Since then, we’ve taken the technology so far beyond that,” Rosa says. The brain ablation model was the first clinical application, and the trigeminal nerve ablation product is the second.
Commenting on what the limited release of the trigeminal nerve ablation system means for the company, Rosa says, “I think it was further validation in that we’re not just a one-product company. We can leverage this technology for use in other areas.”
For example, there is interest in using the system to find new treatment for back pain.
More on Rosa and NeuroOne
Rosa says he is “one of those guys who has been involved in everything” in the medical world, from orthopedics to wound care, from urology to the cardiovascular field—and now, neurology.
His quarter-of-a-century experience leading early-stage medical device companies brought him to NeuroOne, where he has been since the beginning in 2017.
Zimmer Biomet, an Indiana-based medical device company which is also an investor in NeuroOne, has held the exclusive global distribution rights for OneRF since July 2020. NeuroOne ships products to Zimmer Biomet, and then Zimmer sells the product to neurosurgeons and hospitals.
NeuroOne has 17 employees. In the previous fiscal year, the public company generated $9 million in sales, up from under $2 million the fiscal year before. “We showed some significant growth there with just this one technology,” says Rosa.
Sales primarily come from the original OneRF brain ablation system. In April, Rosa says the company has new sites lined up where the new trigeminal neuralgia system will be introduced.
In the current fiscal year, the company expects sales at a minimum of $10.5 million—about a 17% increase over the prior year—which Rosa says is a conservative estimate.
“This company is growing in leaps and bounds, but if you put a number out there, you better hit it,” Rosa says. “If you don’t, you’re going to pay for it.”