Feds OK HistoSonics’ Tumor-Destroying Tech
With federal approval secured earlier this month, Plymouth-based medical startup HistoSonics aims to bring its tumor-destroying ultrasound technology to hospitals and health systems across the nation.
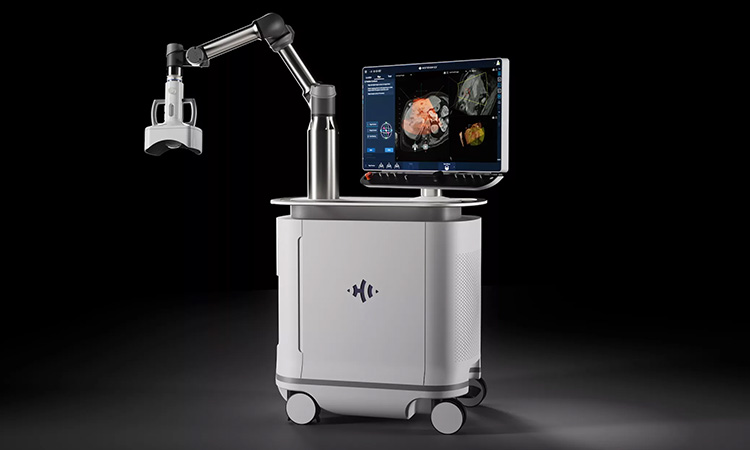
On Oct. 6, the U.S. Food and Drug Administration officially granted HistoSonics authorization to begin marketing its Edison technology, which uses focused ultrasound to destroy liver tumors. Technically speaking, HistoSonics earned the FDA’s “de novo classification,” which gives a company permission to sell novel products that don’t yet exist in the market. The designation differs from the oft-used 510(k) classification process, which is for products that already have analogues in the market.
In a statement issued Oct. 9, HistoSonics president and CEO Mike Blue said the de novo designation is the “most meaningful milestone to date and represents over two-decades of tireless efforts.”
The move will pave the way for HistoSonics to begin selling the Edison system to health systems around the country. The company bills the technology as a non-invasive alternative for liver tumors that might not be feasible to remove with traditional surgery. The process is formally known as “histotripsy,” which is the use of “focused sound energy to produce controlled acoustic cavitation that mechanically destroys and liquifies targeted liver tissue,” according to the company.
According a 2022 paper on HistoSonics’ technology, the liquified tissue is absorbed by the body within 1-2 months. “Histotripsy has also been shown to stimulate an immune response and induce abscopal effects in animal models, which may have positive implications for future cancer treatment,” the paper’s authors noted in an abstract.
HistoSonics’ device is now the “first and only histotripsy platform available in the United States,” company officials said.
The Edison system emerged out of University of Michigan research dating back to 2001. HistoSonics’ main corporate office is in Plymouth, but the company still maintains an office in Ann Arbor, Michigan. The company’s technology earned “breakthrough device designation” from the FDA in 2021.
In 2022, TCB included the Edison system on its Tech 20 list of notable technology achievements from Minnesota companies.