Spineology’s Latest Funding Round Tops $10M
Oakdale-based Spineology Inc., a maker of spinal implant devices, has secured more than $10 million in a recent funding round, according to SEC documents published Friday.
Eighty investors contributed in the round in which approximately $10.18 million in debt and option, warrant or the right to acquire other securities from the company were offered.
Spineology began taking investments of $25,000 or more for the recently closed round about a year ago.
Based on filings made by Spineology since its 1997 founding, this was the company’s largest funding round to date. Altogether, the company had raised less than $10 million over several rounds, according Crunchbase, which monitors fundraising efforts by startups.
The company hasn’t yet explained what it will do with the money.
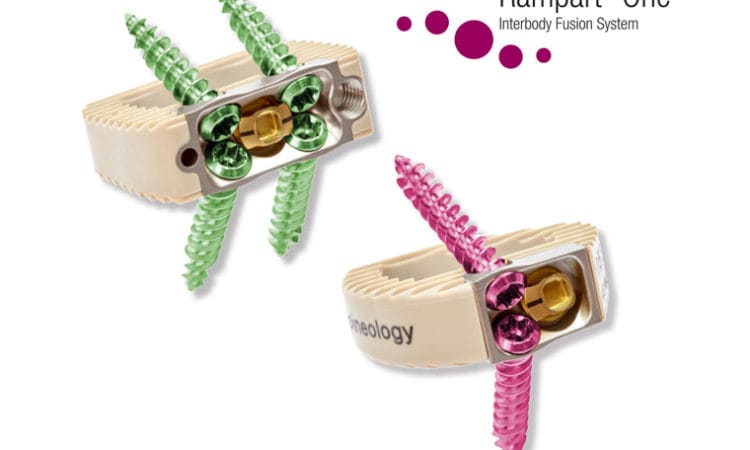
Within the last year, the biotechnology has pushed two products through the FDA approval process. Its Rampart One device, which screws into the lumbar section at the end of the spine, was cleared for sale to hospitals in the U.S. last June. Meanwhile, last October, its Rampart Duo implant received FDA approval. The device is used to maintain sagittal alignment (or a front-to-back imbalance in the spine) during surgery.
Spineology claims to bring in revenues of at least $25 million but no more than $100 million annually, it said in its SEC filing. Chief executive John Booth, the former CEO of Phillips Plastics Corp. and Microvena Corp., has led the company since 2004 and had been a member of Spineology’s board since its inception.