The Medical Device Sterilization Plant Next Door
The federal government is cracking down on a widely used sterilization agent for medical devices, drawing concern from the medtech industry and cheers from environmental advocates.
Last month, the Environmental Protection Agency issued a new rule aimed at significantly cutting emissions of ethylene oxide, a gas used to sterilize a staggering number of pieces of medical equipment every year. First discovered in the 19th century, ethylene oxide has, for better or worse, become pivotal in the medical device industry. These days, the colorless, odorless gas is used to sterilize as many as 20 billion medical devices a year – everything from catheters to joint replacements and much more.
But ethylene oxide, or EtO, is also a carcinogen, a fact that’s largely undisputed among both industry leaders and environmental advocates. The Biden administration is looking to clamp down on it as part of its wider “cancer moonshot” initiative. The EPA’s rule calls for cutting emissions of ethylene oxide by 90%.
Bobbie Dressen, president of the Golden Valley-based health-tech trade group Medical Alley, said that pretty much every surgery performed in U.S. hospitals will use one or more devices sterilized by the gas. On March 14, the same day that the EPA released its new final rule on ethylene oxide, Medical Alley responded with a statement expressing concern. The agency’s new rule, Dressen said, could “jeopardize patient access to care.”
Based on her conversations with sterilization facilities, Dressen said that the timing of the EPA’s new ruling is simply untenable. The agency is giving sterilization facilities at least two years to get into compliance, but they say that’s not enough.
“It’s not a lack of attention or willingness to comply,” Dressen said in a late March interview. “It’s the concern about not being able to accomplish it in those time periods, given the fact that to date, there have been no alternatives” ready to roll out on a quick, mass scale.
Medical sterilization companies also warn that the new rule could create bottlenecks in the nation’s already fragile medical supply chain. That’s because the rule would necessitate a host of operational changes, and investments. Dressen said those would lead to cost increases that would ultimately funnel down to consumers.
“In this age of trying to reduce medical care costs,” she said, “these additional things are now adding layers to that cost.”
Dressen and others in the industry agree that it’s worth exploring alternatives to ethylene oxide. But the timing of the EPA’s rule is their biggest concern.
Meanwhile, two medical device giants with a significant Minnesota presence said they’re “evaluating” the EPA’s new requirements.
“We are aware of the EPA’s recent ruling on ethylene oxide requirements for commercial sterilizers,” a Medtronic spokesperson said in an email. “We are evaluating these new requirements and will determine next steps.”
A Boston Scientific spokesperson said much the same: “We are evaluating the potential effects of the new rules and requirements for compliance and are not commenting further at this time.”
A long-simmering concern
At the same time, environmental advocates note that the EPA has been weighing how to handle the gas for many years now. Back in 2005, the agency even considered banning the use of ethylene oxide outright at any new sterilization facilities. The EPA ultimately didn’t enact that ban, but environmentalists maintain that the industry should have been prepared for a crackdown at some point.
“There have been a number of points in the last 20 years in which there have been flags about this substance,” said Darya Minovi, a senior analyst with the Center for Science and Democracy at the Union of Concerned Scientists, a D.C.-based advocacy group. “This is not new or novel information.”
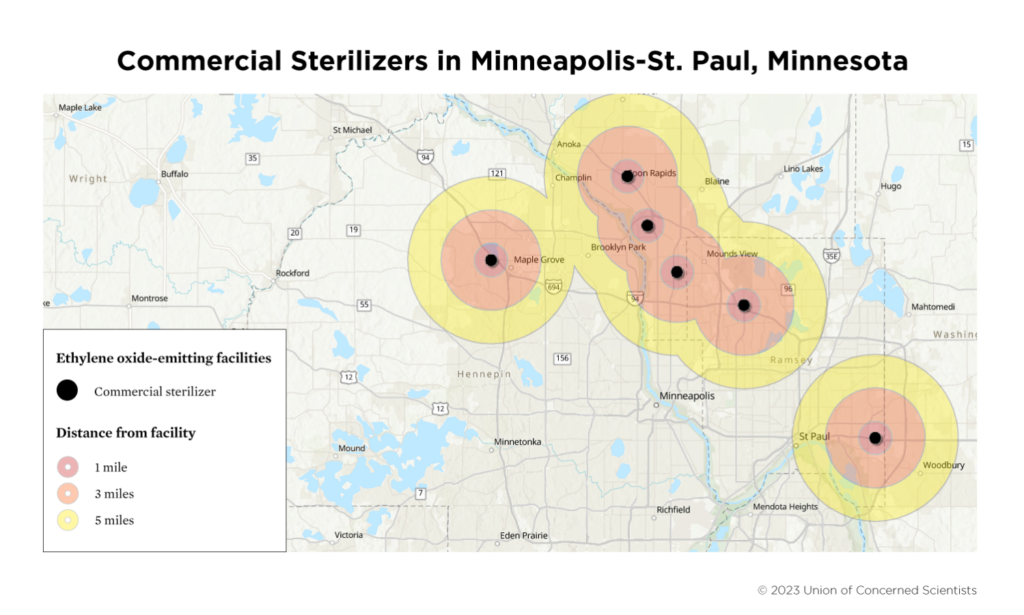
Last year, Minovi analyzed the geographical distribution of the 88 medical sterilization plants in the U.S. that currently use ethylene oxide. At least six are in Minnesota, and most are in the northerly suburbs of Minneapolis. Medtronic and Steris have two plants in Coon Rapids, for instance. While U.S. Census data show Coon Rapids is 77% white, Minovi pointed out that these plants are often located close to communities predominantly composed of people of color.
“These facilities often operate near marginalized communities,” the Union of Concerned Scientists said in a February 2023 statement. “The 5-mile radius around these sites include 14.2 million people. Of these people, nearly 60 percent identify as people of color, 31 percent are low-income, and 8 percent have limited English language proficiency.”
In late March, ABC News’ Nightline newsmagazine reported on ethylene oxide plants in a Memphis neighborhood where many Black residents live. Before releasing the March 14 rule, EPA did outreach in Memphis and many other communities with sterilization plants.
While ethylene oxide is a known carcinogen, the precise cancer risk for folks living in communities with sterilization plants isn’t entirely clear. Minovi’s analysis didn’t specifically look into cancer rates near these plants; rather, her study was a “proximity analysis.”
“It doesn’t mean that someone who lives right between two facilities is absolutely going to be exposed to hazardous levels of ethylene oxide, but we wanted folks to be aware if there were multiple facilities in their community,” she said.
In its final rule, EPA reported that, based on a 2016 analysis, “EtO is a far more potent carcinogen than we had understood.”
“We have determined that approximately 23 of these facilities pose high lifetime cancer risks to the surrounding communities,” the agency said in the ruling.
Plus, there are inherent risks to operating a facility with carcinogenic chemicals inside. Accidents can happen.
“If there were a leak, or if they had a failure in a facility’s chamber, it could release a very toxic and carcinogenic gas,” said Natalia Tretyakova, a chemical toxicologist and professor of medicinal chemistry at the University of Minnesota’s College of Pharmacy. “I’m not concerned that’s happening right now, or that they’re releasing it all the time. But there is a risk.”
Tretyakova said that if ethylene oxide reacts with DNA, it can cause mutations that lead to cancer. She said there’s “ample evidence” that industrial exposures to the gas can cause blood cancers like leukemia. “There are safer alternatives,” she said. Radiation is one. And just this year, the U.S. Food and Drug Administration approved vaporized hydrogen peroxide as a way to sterilize medical devices.
Tretyakova ultimately believes that at some point the future, companies won’t be using ethylene oxide. She pointed out that the gas has already been prohibited in sterilization of foods in Europe.
In an email, a Medical Alley spokesperson acknowledged that “radiation is, indeed, an alternative method of sterilization.”
“It is an appropriate method for certain types of devices,” the spokesperson said of radiation. “Any effective method of sterilization, however, is dependent upon the design of the device, and its packaging. For the majority of devices, EtO provides the best method to ensure the performance, sterility, safety, and effectiveness of the device.”
There’s also a winding road to validating a new sterilization procedure. The FDA, for instance, would need to greenlight any changes.
“The industry continues research to develop alternatives to EtO,” Medical Alley said. “Some have been identified. But none that offer the scale and applicability for a wide variety of devices, as does EtO.”
For her part, Minovi with the Union of Concerned Scientists said that her group isn’t advocating for a “cold-turkey ban of EtO at this time.”
“Our medical supply chain has, for better or worse, been built around this substance that’s very effective at killing microbes,” she said. “But there are other substances that we can use.”








