Bloomington Med Tech Startup Wants to Save Sepsis Patients with a Breath Test
Tom Burke, founder of Vail Scientific, a Bloomington-based med tech startup focused on early sepsis detection, noticed a problem inside hospitals.
Each year, at least 1.7 million adults in the United States develop sepsis, a medical emergency that occurs when an infection—bacterial, viral, fungal, or parasitic—causes the immune system to attack its own tissues anywhere in the body. More than 350,000 adults who develop the disease die during hospitalization or are discharged to hospice care. The No. 1 cause of sepsis is a urinary tract infection. When the infection starts to shut down organs, it’s called septic shock.
Patients with sepsis can be in waiting rooms for three to four hours, Burke says, “which is particularly dangerous with this disease, because the mortality rate goes up 8% each hour.”
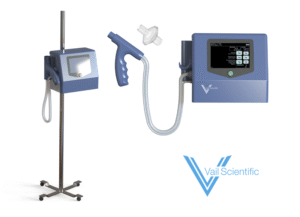
Vail Scientific, founded in 2015, is aiming to avert as many as 80% of sepsis deaths via rapid diagnosis and treatment, to be achieved with a new device called VSNO, which stands for vital signs and nitric oxide. It is a battery-powered tool the size of a desk telephone that mounts onto an IV pole and monitors a patient’s breath for a chemical called nitric oxide. This way, Vail Scientific claims, its new device can detect sepsis within two minutes.
Burke imagines hospitals using the device on patients during triage in emergency departments.
“If you can catch that patient much earlier and treat them, the chance of having a better outcome for the patient is significant,” Burke explains, and can save the patient and hospital “lots of money” by avoiding an ICU stay.
Current Sepsis Detection
One way physicians screen for sepsis is by counting white blood cells. This can take up to three hours, and by the time those results are finalized, the patient may be dead, as sepsis’ mortality rate increases cumulatively by the hour.
Vail Scientific’s VSNO tests four vitals: heart rate, body temperature, respiratory rate, and nitric oxide.
When the body detects something foreign, it releases nitric oxide to dilate blood vessels and destroy toxins in the blood. [Too much or too little nitric oxide signals sepsis.]
There is not a great way to measure nitric oxide in the blood, because its half-life is mere seconds. “By the time you take a blood draw and try to run to the lab, it’s gone,” Burke explains.
RELATED: How Medica Is Navigating Massive Health Care Challenges
VSNO measures nitric oxide from an exhale. “This tool gives the doctor a risk score and says, ‘OK, this is someone you need to get back in the ED [emergency department] right away,” he says.
The device is still on its way to commercialization, as Vail Scientific has completed three successful trials at the University of Minnesota and M Health partners.
Progress as a Startup
Burke founded Vail Scientific more than a decade ago, following leadership roles with other pharmaceutical and startup companies, such as Upsher-Smith Laboratories in Maple Grove. The startup operates in a room with three desks inside a Bloomington building that also houses dozens of executive offices.
Vail Scientific’s four founders knew only that they wanted to solve a life-or-death problem in the health care system.
“There’s great potential, and we are doing something that’s going to change people’s lives once this gets to market,” Burke says.
During its seed funding round, Vail Scientific raised $3.3 million. It has raised an additional $4 million in Series A funding. Burke has expanded its staff to six employees, and the startup has multiple patents pending.
On the national level, Vail Scientific is one of three Minnesota med tech startups getting spotlighted on the MedTech Startup Innovation Stage at Device Talks, a networking event series for the medtech industry, on Monday. The event is at the McNamara Alumni Center at the University of Minnesota-Twin Cities.
The next step for Vail Scientific is to submit the device to a pre-submission trial for FDA clearance, a process that can take a couple years, Burke says. Burke hopes VSNO could be used in ambulances and nursing homes, as well.