Mayo Makes Third Investment in Digital Heart Health Firm
For the third time in less than a year, the Mayo Clinic has made a financial investment in AliveCor, a Silicon Valley digital health startup that is commercializing Mayo-developed technology to detect heart health risks via a smartphone app.
The two parties first revealed a research collaboration and equity stake in November, then followed that up in March when Mayo participated in a $30 million Series D venture capital round for AliveCor led by Omron Healthcare.
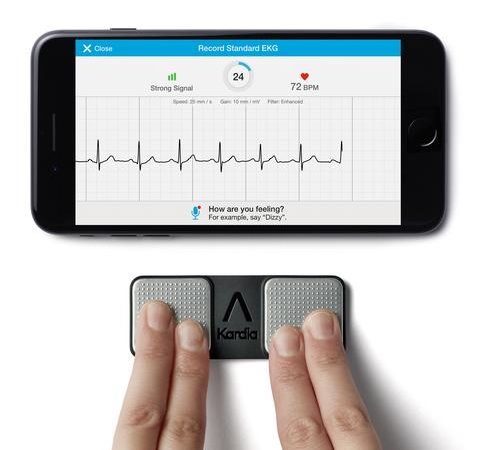
Both were in support of the company’s Kardia Mobile product, which allows consumers to generate a “clean” clinical-level electrocardiogram (ECG) by placing their fingers on a small pad connected to a smartphone. The ECG can be uploaded to a secure cloud and the signals analyzed using Mayo’s expertise in machine learning algorithms to detect tiny changes in blood potassium levels – in essence, hidden early indicators of enhanced risk for cardiac arrhythmia such as atrial fibrillation, as well as for heart disease and stroke.
Now, Mayo and AliveCor are further expanding their relationship. They announced in a statement released last week they are working together to expand the capabilities of Kardia Mobile to address risks for long QT syndrome (LQTS), a “sudden death” disorder best known for fatally striking seemingly healthy children and young adults while they are playing sports.
LQTS is characterized by sudden, massive cardiac arrest. It usually goes undiagnosed; the new collaboration is meant to change that reality as a step toward the first universal screening for the condition, which is “easily preventable” if detected early.
Along with a research collaboration aimed at gaining U.S. Food and Drug Administration approval for the LQTS use, the parties announced an unspecified follow-on financial “investment” by Mayo—its third overall in Mountain View, California-based AliveCor.
Like with the previous partnership, the LGTS effort will combine AliveCor's artificial intelligence technology with Mayo's patented algorithms. They were developed by Dr. Paul Friedman, the clinic’s vice chair of cardiovascular medicine and director of its implantable device lab; Dr. Michael Ackerman, director of Mayo’s Sudden Death Genomics Laboratory; and several others.
LQTS is both a congenital and acquired disorder. The inherited form affects 160,000 people in the United and causes 3,000 to 4,000 sudden deaths in children and young adults each year. It is estimated to affect about 1 in 7,000 people — but no one knows for sure because it often goes undiagnosed, according to the National Institutes of Health.
The agency says that while unexplained sudden deaths in children are rare, LQTS often is the cause when they do occur. Inherited LQTS usually is first detected during childhood or young adulthood. Half of all people who have LQTS experience the first abnormal heart rhythms by age 12, with 90 percent by age 40. The condition rarely is diagnosed after that.
LQTS is also more common in women than men. Women who have LQTS are more likely to faint or die suddenly from the disorder during menstruation and shortly after giving birth. Additionally, children who are born deaf also are at increased risk for LQTS.
Dr. Ackerman’s research at Mayo has been focused on using genomics to identify people at greatest risk of sudden death conditions such as LQTS and Sudden Infant Death Syndrome. He said last week’s statement that the new collaboration could be a key development in preventing LQTS.
“This agreement makes our vision of universal screening for the early detection of long QT syndrome—a potentially lethal, yet highly treatable condition—one step closer to reality,” it read. “The electrical heart cycle is emerging as the next vital sign. With very few exceptions, we now know that a prolonged cycle—whether caused by genetics, drugs, electrolyte disturbances or by other diseases—indicates increased risk for early death.
“Any of these deaths could be averted with simple preventive and/or counteractive measures.”